Why Insulin Resistance May Accelerate Aging
Of the modifiable biological liabilities that accelerate aging, insulin resistance is among the most prevalent, most measurable, and most consistently underdetected in standard annual panels. A fasting glucose within the "normal" range does not mean insulin resistance is absent. It often means the pancreas is compensating hard enough to maintain glucose at the cost of chronically elevated insulin.
This post explains what insulin resistance is, how it accelerates biological aging through multiple pathways, and why it belongs at the center of any serious longevity protocol. It connects to the GLP-1 longevity post GLP-1s and Longevity and the metabolic optimization discussion Weight Loss vs Muscle Loss: The GLP-1 Problem Nobody Talks About.
What Insulin Resistance Actually Is
Insulin is a signaling hormone. Its primary role is to move glucose from the bloodstream into cells, particularly muscle and liver cells, where it is stored or burned for energy. Insulin resistance occurs when cells stop responding efficiently to insulin's signal. The pancreas compensates by producing more insulin to achieve the same glucose disposal effect.
The result is chronically elevated insulin (hyperinsulinemia) in the presence of what may appear to be normal blood glucose. Standard blood panels measure fasting glucose. They rarely measure fasting insulin. This means insulin resistance often goes undetected until it has progressed to pre-diabetes or type 2 diabetes, by which point years of compounding metabolic damage have already occurred.
Measuring fasting insulin alongside glucose and calculating HOMA-IR gives a far more accurate picture of insulin sensitivity than glucose alone. This is standard practice in any serious biological audit. It is not in most annual physicals.
How Insulin Resistance Accelerates Aging
Visceral fat accumulation
Insulin resistance promotes the preferential storage of fat around the organs, visceral adipose tissue, rather than subcutaneous fat. Visceral fat is metabolically active: it secretes inflammatory cytokines that worsen insulin resistance and drive systemic inflammation. This creates a reinforcing cycle that is difficult to break without targeted intervention.
Chronic inflammation
Hyperinsulinemia activates inflammatory pathways including NF-kB. Chronic low-grade inflammation is one of the most consistent drivers of biological aging across organ systems. The term "inflammaging" reflects this relationship, and it is not metaphorical. Elevated hs-CRP and IL-6 in an executive's panel are often downstream of undetected insulin resistance.
Mitochondrial dysfunction
Insulin resistance impairs glucose uptake in cells, forcing a shift in metabolic fuel use. Over time, this contributes to mitochondrial inefficiency, less efficient energy production, more oxidative stress, and accelerated cellular aging. Declining energy levels in a high-performing executive are often a mitochondrial signal before they are anything else.
Cardiovascular risk
Elevated insulin promotes arterial smooth muscle cell proliferation, lipid dysregulation (elevated triglycerides, low HDL, increased small dense LDL), and endothelial dysfunction. These are the substrate of atherosclerosis. Cardiovascular disease is not a disease that appears suddenly in your 60s. It is an insulin-resistance problem that compounds quietly over decades.
Hormonal disruption
Insulin interacts with sex hormone-binding globulin (SHBG), affecting testosterone and estrogen availability. Chronically elevated insulin suppresses SHBG, altering the hormonal environment in ways that affect body composition, cognitive performance, and recovery capacity. Male executives with unexplained low-normal testosterone should have insulin measured before anything else.
Cognitive risk
The brain is an insulin-sensitive organ. Insulin resistance in brain tissue impairs neuronal glucose metabolism and has been associated with increased risk of cognitive decline. The relationship between metabolic dysfunction and neurodegeneration is one of the most active areas in aging research.
Why "Normal" Fasting Glucose Is Not Enough
The standard system flags insulin resistance at fasting glucose above 100 mg/dL (pre-diabetes) or 126 mg/dL (diabetes). By those cutoffs, insulin resistance has often been progressing for years.
Optimal fasting insulin is below 5 mIU/L. HOMA-IR below 1.0 reflects good insulin sensitivity. Many individuals with fasting glucose in the 80s have HOMA-IR values indicating early insulin resistance, particularly those with high visceral fat, sedentary behavior, or poor sleep quality.
At Diab Longevity, fasting insulin and HOMA-IR are standard components of the biological audit. They are not optional add-ons. Metabolic age cannot be accurately assessed without them.
What Actually Moves Insulin Sensitivity
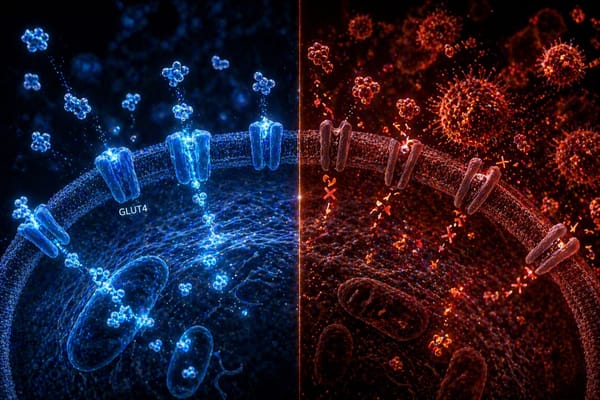
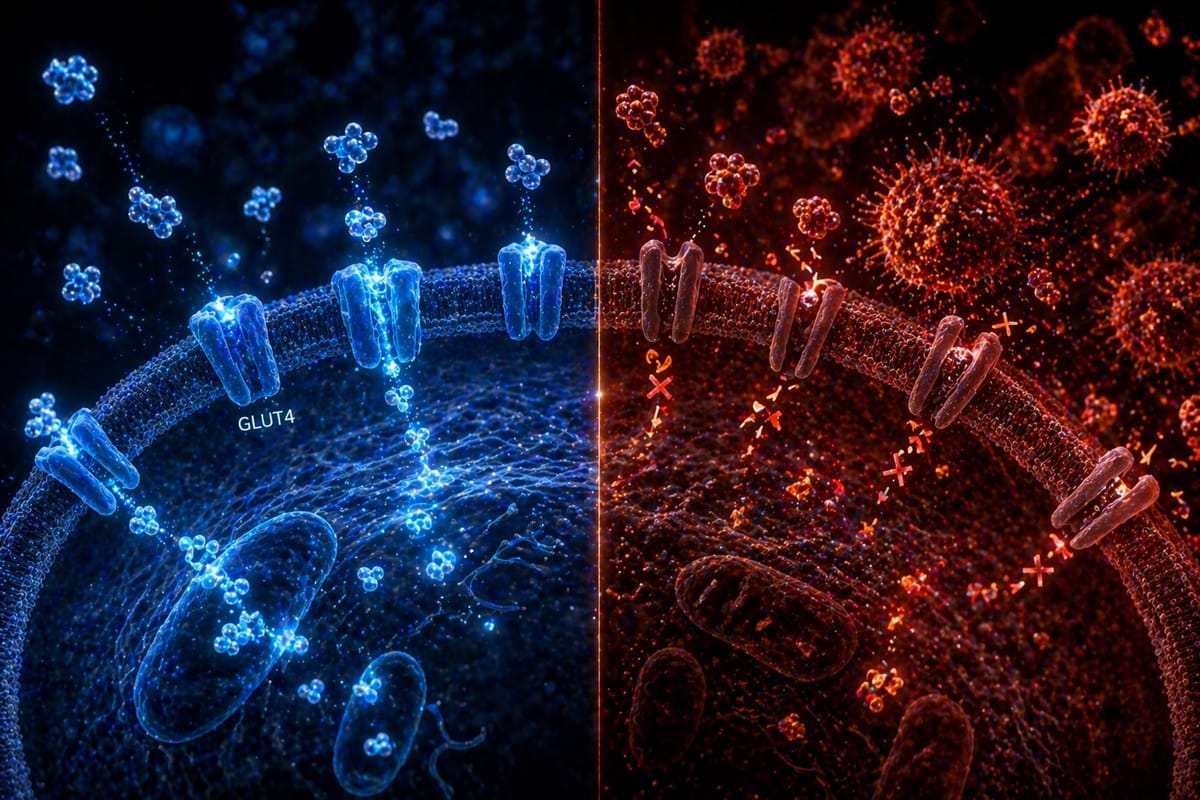
The most effective tools for improving insulin sensitivity are well characterized: progressive resistance training (which increases GLUT4 expression in skeletal muscle), dietary modification particularly around processed carbohydrates and meal timing, sleep quality optimization, visceral fat reduction, and where clinically indicated, pharmacological support including GLP-1 agonists and metformin.
These interventions are quantifiable. Their effects on insulin sensitivity can be tracked in real time. This is exactly the kind of biological liability DiabOS is designed to monitor and respond to.
Start Your Optimization Review.
*Medical disclaimer: This content is educational and does not constitute medical advice. All protocols are individualized and supervised by a licensed physician.*